Integrated Genetic, Molecular, and Wearable Sensor Biomarkers Enable Bayesian Machine Learning-Driven Precision Stratification in Parkinson's Disease
Tirhekar, H.M., Yadav, P., Bajaj, C.
A Comprehensive Multi-Cohort Validation Study
View full paper →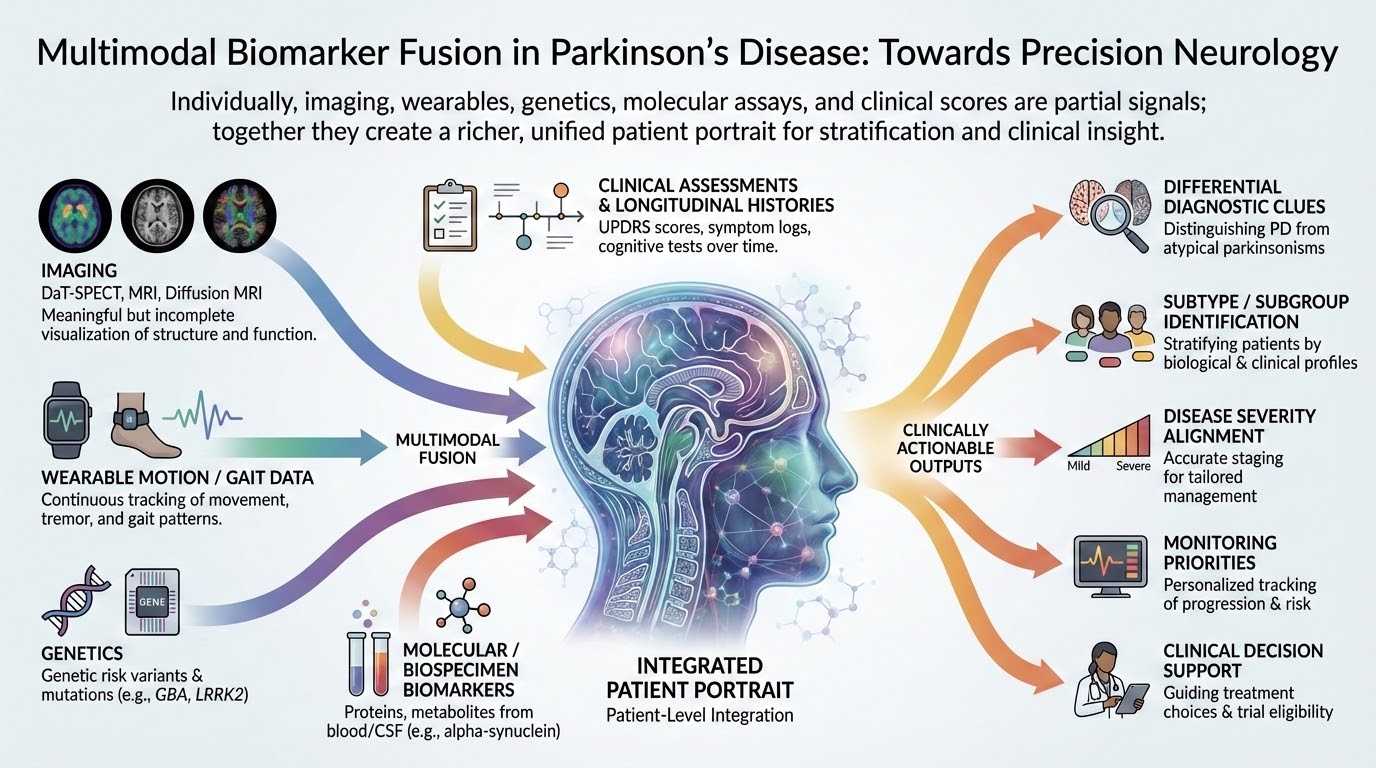
Multimodal biomarker fusion: imaging, wearables, genetics, molecular assays, and clinical scores converge into a unified patient portrait yielding clinically actionable outputs.
Key findings
- LRRK2 G2019S carriers show measurably higher motor severity, enabling genetic risk stratification and personalized prognostic counseling.
- Wearable arm-swing asymmetry detects unilateral substantia nigra degeneration; dual-task gait cost reveals cognitive-motor network failure.
- Bayesian clustering with Dirichlet Process priors identifies four motor phenotypes with transparent model selection via Evidence Lower Bound.
- A risk prediction model enables personalized prognostic counseling and treatment selection across genetic and molecular subtypes.
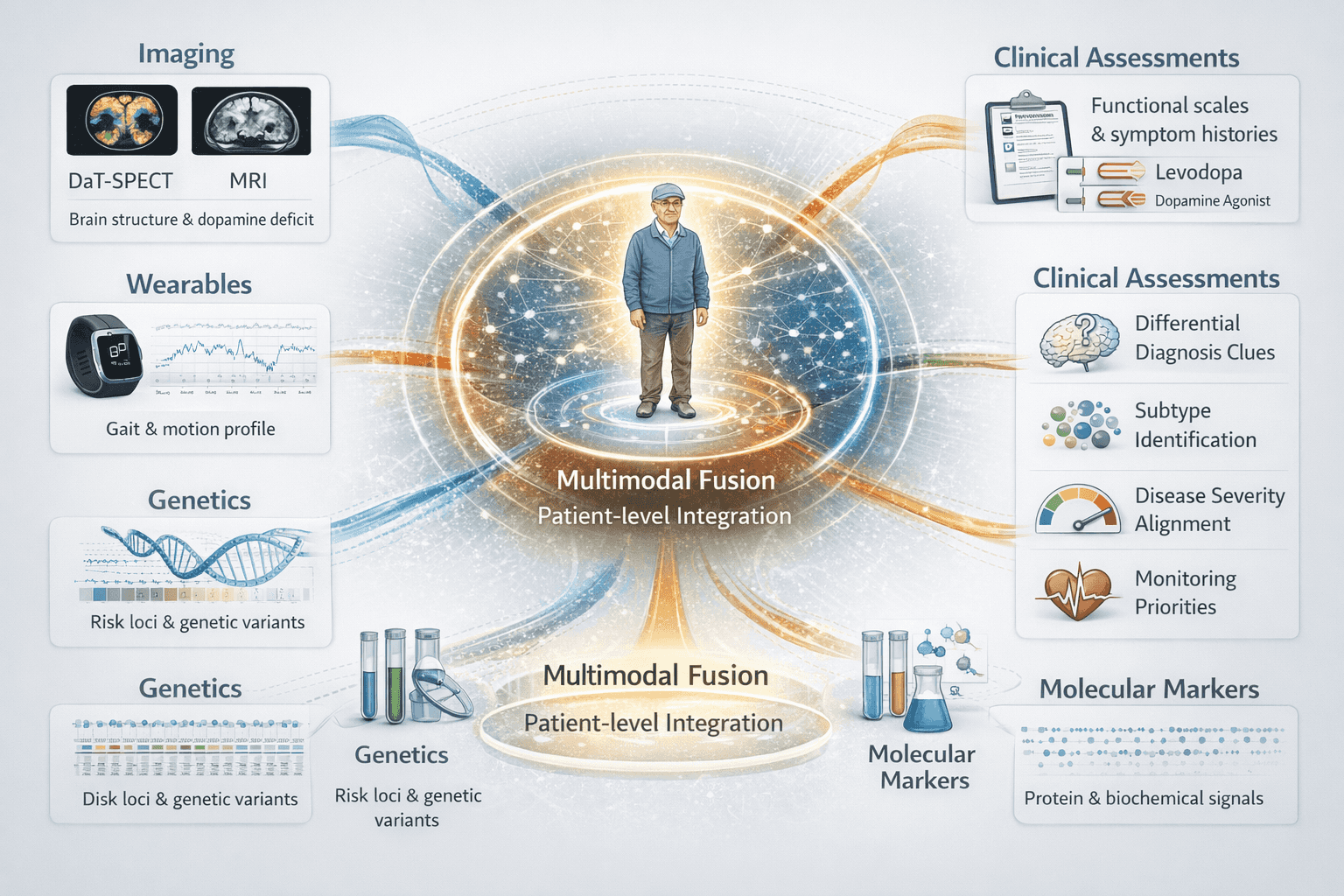
Patient-level multimodal fusion: imaging, wearables, genetics, and molecular markers integrated into clinical assessment outputs including differential diagnosis clues, subtype identification, severity alignment, and monitoring priorities.
Cohorts and methods
This study leverages two large complementary cohorts: the Parkinson's Progression Markers Initiative (PPMI) with 4,775 unique patients and 14,473 longitudinal assessments spanning 14.5 years, and the LRRK2 Consortium with 627 individuals (347 G2019S carriers, 280 non-carrier controls).
The framework integrates genetic profiling (LRRK2 G2019S genotyping), molecular biomarkers (urinary phospho-S1292-LRRK2, CSF alpha-synuclein seed amplification), wearable phenotyping (tri-axial IMU accelerometers and gyroscopes at 100 Hz), and clinical assessments (MDS-UPDRS, MoCA, UPSIT, SCOPA-AUT) through Bayesian Gaussian Mixture Models with uncertainty quantification.
Clinical outputs
Genetic risk stratification
Personalized counseling based on LRRK2 carrier status and associated motor severity profiles.
Continuous digital monitoring
Wearable-derived gait and arm-swing metrics for ecological, day-to-day tracking beyond clinic visits.
Mechanism-based targeting
Molecular biomarker profiles guide selection of pathway-specific therapeutic strategies.
Prodromal detection
Early intervention windows identified during pre-manifest stages (Braak stages 1-3).